The relatively new antibiotic cefiderocol given in the form of eye drops may be a way to combat a type of ocular infection that broke out in the United States last year, according to research presented at the 2024 annual meeting of the Association for Research in Vision and Ophthalmology (ARVO).
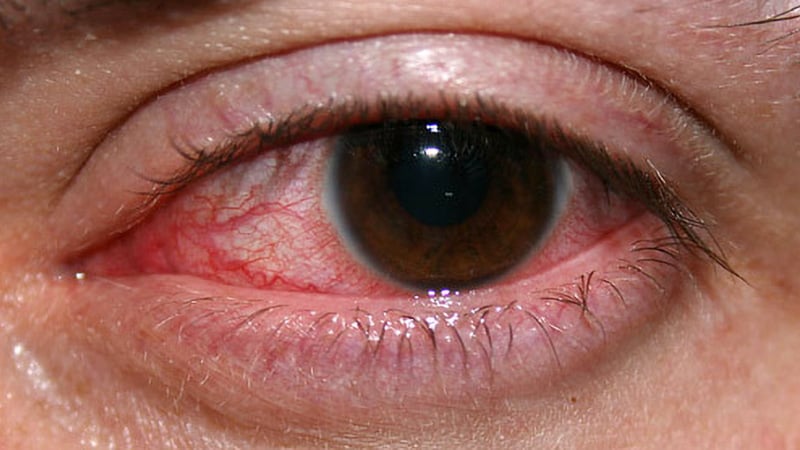
The infections, linked to contaminated bottles of artificial tears, were detected in 81 patients in 18 states. The outbreak led to loss of vision in 14 patients, surgical removal of the eyeball in four patients, and four deaths, according to health officials.
An extensively drug-resistant strain of Pseudomonas aeruginosa that had not previously been reported in the country caused the infections. Scientists cautioned last year that the bacteria potentially could spread from person to person.
At ARVO on May 6, Eric G. Romanowski, MS, research director of the Charles T. Campbell Ophthalmic Microbiology Laboratory at the University of Pittsburgh Medical Center, Pittsburgh, Pennsylvania, described studies that his lab conducted evaluating topical cefiderocol as a potential treatment option for these infections (Abstract 2095).
Investigators had found that the bacterial strain was susceptible to this medication, which was approved by the US Food and Drug Administration in 2019 as a treatment for complicated urinary tract infections. But the antibiotic had not been tested as an eye drop.
“We showed that the ‘Trojan-horse’ antibiotic, cefiderocol…was non-toxic and effective against the highly resistant outbreak strain in an experimental model of infection,” Romanowski and co–lead investigator Robert M. Q. Shanks, PhD, said in a statement about their research. “These results demonstrate that topical cefiderocol could be a new weapon in the ophthalmologist’s arsenal for the treatment of corneal infections caused by highly antibiotic-resistant Pseudomonas aeruginosa.”
Experimental Models
Romanowski’s group, with colleagues at the Geisel School of Medicine at Dartmouth University, Hanover, New Hampshire, used minimum inhibitory concentration testing to evaluate the effectiveness of cefiderocol against 135 isolates from eye infections. They also tested ocular toxicity and antibiotic efficacy of cefiderocol eye drops in a rabbit model of keratitis caused by the bacterial strain.
Cefiderocol was “well tolerated on rabbit corneas,” they reported. It also was effective in vitro against the isolates and in vivo in the rabbit model of keratitis.
They first published their findings as a preprint in September 2023 and then in Ophthalmology Science in December.
A ‘Duty to the Profession’
Their paper noted that “there is no current consensus as to the most effective antimicrobial strategy to deal with” extensively drug-resistant keratitis.
During the outbreak, clinicians tried various treatment regimens, with mixed results. In one case, a combination of intravenous cefiderocol and other topical and oral medications appeared to be successful.
Romanowski’s team decided to test cefiderocol drops with their own resources “as a duty to the profession,” he said. “Not many labs do these types of studies.”
“We would like to see further development of this antibiotic for potential use,” Romanowksi added. “It would be up to any individual clinician to determine whether to use this antibiotic in an emergency situation.”