Avoid Unapproved Potassium Phosphates Injection in Pediatric Patients
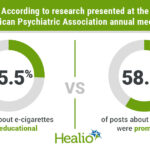
The FDA has issued a safety alert regarding the use of unapproved potassium phosphates drug products by Hospira in pediatric patients. These products may result in daily aluminum exposures exceeding recommended limits, potentially leading to various health issues. To prevent this, the FDA recommends using alternative products such as Fresenius Kabi’s or CMP Development’s Potassium…