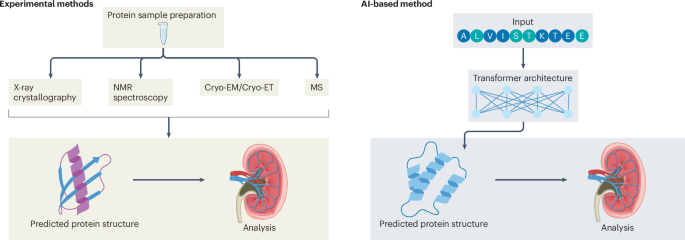
Summarize this content to 100 words: Balzer, M. S., Rohacs, T. & Susztak, K. How many cell types are in the kidney and what do they do? Ann. Rev. Physiol. 84, 507–531 (2022).Article
CAS
Google Scholar
Lewis, S. et al. “SLC-omics” of the kidney: solute transporters along the nephron. Am. J. Physiol. Cell Physiol. 321, C507–C518 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Hoenig, M. P. & Zeidel, M. L. Homeostasis, the milieu interieur, and the wisdom of the nephron. Clin. J. Am. Soc. Nephrol. 9, 1272–1281 (2014).Article
CAS
PubMed
PubMed Central
Google Scholar
Hansen, J. et al. A reference tissue atlas for the human kidney. Sci. Adv. 8, eabn4965 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Tomas, N. M., Mortensen, S. A., Wilmanns, M. & Huber, T. B. Across scales: novel insights into kidney health and disease by structural biology. Kidney Int. 100, 281–288 (2021).Article
CAS
PubMed
Google Scholar
Beck, M., Covino, R., Hänelt, I. & Müller-McNicoll, M. Understanding the cell future views of structural biology. Cell 187, 545–562 (2024).Article
CAS
PubMed
Google Scholar
Hipp, M. S., Park, S. H. & Hartl, F. U. Proteostasis impairment in protein-misfolding and -aggregation diseases. Trends Cell Biol. 24, 506–514 (2014).Article
CAS
PubMed
Google Scholar
Gámez, A. et al. Protein misfolding diseases: prospects of pharmacological treatment. Clin. Genet. 93, 450–458 (2018).Article
PubMed
Google Scholar
Ramazi, S. & Zahiri, J. Post-translational modifications in proteins: resources, tools and prediction methods. Database 7, baab012 (2021).Article
Google Scholar
Peters, A., Nawrot, T. S. & Baccarelli, A. A. Hallmarks of environmental insults. Cell 184, 1455–1468 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Liu, Z. Z., Yang, J., Du, M. H. & Xin, W. Functioning and mechanisms of PTMs in renal diseases. Front. Pharmacol. 14, 1238706 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Dubin, R. F. et al. Proteomics of CKD progression in the chronic renal insufficiency cohort. Nat. Commun. 14, 6340 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Jha, V. et al. Global economic burden associated with chronic kidney disease: a pragmatic review of medical costs for the inside CKD research programme. Adv. Ther. 40, 4405–4420 (2023).Article
PubMed
PubMed Central
Google Scholar
Karplus, M. & Weaver, D. L. Protein-folding dynamics. Nature 260, 404–406 (1976).Article
CAS
PubMed
Google Scholar
Daggett, V. & Fersht, A. The present view of the mechanism of protein folding. Nat. Rev. Mol. Cell Biol. 4, 497–502 (2003).Article
CAS
PubMed
Google Scholar
Dill, K. A., Ozkan, S. B., Shell, M. S. & Weikl, T. R. The protein folding problem. Annu. Rev. Biophys. 37, 289–316 (2008).Article
CAS
PubMed
PubMed Central
Google Scholar
Englander, S. W. & Mayne, L. The nature of protein folding pathways. Proc. Natl Acad. Sci. USA 111, 15873–15880 (2014).Article
CAS
PubMed
PubMed Central
Google Scholar
Levinthal, C. Are there pathways for protein folding. J. Chim. Phys. 65, 44–45 (1968).Article
Google Scholar
Louros, N., Schymkowitz, J. & Rousseau, F. Mechanisms and pathology of protein misfolding and aggregation. Nat. Rev. Mol. Cell Biol. 24, 912–933 (2023).Article
CAS
PubMed
Google Scholar
Anfinsen, C. B. Principles that govern the folding of protein chains. Science 181, 223–230 (1973).Article
CAS
PubMed
Google Scholar
Kresge, N., Simoni, R. D. & Hill, R. L. The thermodynamic hypothesis of protein folding: the work of Christian Anfinsen. J. Biol. Chem. 281, E11–E13 (2006).Article
CAS
Google Scholar
Pusey, M. L. et al. Life in the fast lane for protein crystallization and X-ray crystallography. Prog. Biophys. Mol. Biol. 88, 359–386 (2005).Article
CAS
PubMed
Google Scholar
Perutz, M. F. et al. Structure of haemoglobin: a three-dimensional Fourier synthesis at 5.5-A. resolution, obtained by X-ray analysis. Nature 185, 416–422 (1960).Article
CAS
PubMed
Google Scholar
Kendrew, J. C. et al. A three-dimensional model of the myoglobin molecule obtained by X-ray analysis. Nature 181, 662–666 (1958).Article
CAS
PubMed
Google Scholar
Bax, A. & Clore, G. M. Protein NMR: boundless opportunities. J. Magn. Reson. 306, 187–191 (2019).Article
CAS
PubMed
PubMed Central
Google Scholar
Wüthrich, K. NMR studies of structure and function of biological macromolecules (Nobel Lecture). J. Biomol. NMR 27, 13–39 (2003).Article
PubMed
Google Scholar
Henderson, R. Realizing the potential of electron cryo-microscopy. Q. Rev. Biophys. 37, 3–13 (2004).Article
CAS
PubMed
Google Scholar
Henderson, R. From electron crystallography to single particle cryo-EM (Nobel Lecture). Angew. Chem. Int. Ed. 57, 10804–10825 (2018).Article
CAS
Google Scholar
Frank, J. Single-particle reconstruction of biological molecules — story in a sample (Nobel Lecture). Angew. Chem. Int. Ed. 57, 10826–11084 (2018).Article
CAS
Google Scholar
Dubochet, J. On the development of electron cryo-microscopy (Nobel Lecture). Angew. Chem. Int. Ed. 57, 10842–10846 (2018).Article
CAS
Google Scholar
Beck, M. & Baumeister, W. Cryo-electron tomography: can it reveal the molecular sociology of cells in atomic detail? Trends Cell Biol. 26, 825–837 (2016).Article
PubMed
Google Scholar
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).Article
PubMed
Google Scholar
Liu, Y.-T. et al. Isotropic reconstruction for electron tomography with deep learning. Nat. Commun. 13, 6482 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Kao, A. H. et al. Development of a novel cross-linking strategy for fast and accurate identification of cross-linked peptides of protein complexes. Mol. Cell Proteom. 10, M110.002212 (2011).Article
Google Scholar
Murata, K. et al. Structural determinants of water permeation through aquaporin-1. Nature 407, 599–605 (2000).Article
CAS
PubMed
Google Scholar
Frick, A. et al. X-ray structure of human aquaporin 2 and its implications for nephrogenic diabetes insipidus and trafficking. Proc. Natl Acad. Sci. USA 111, 6305–6310 (2014).Article
CAS
PubMed
PubMed Central
Google Scholar
Rahuel, J., Priestle, J. P. & Grutter, M. G. The crystal-structures of recombinant glycosylated human renin alone and in complex with a transition-state analog inhibitor. J. Struct. Biol. 107, 227–236 (1991).Article
CAS
PubMed
Google Scholar
Arakawa, T. et al. Crystal structure of the anion exchanger domain of human erythrocyte band 3. Science 350, 680–684 (2015).Article
CAS
PubMed
Google Scholar
Yu, B. W., Hu, Z. Z., Kong, D. D., Cheng, C. & He, Y. N. Crystal structure of the CTLD7 domain of human M-type phospholipase A2 receptor. J. Struct. Biol. 207, 295–300 (2019).Article
CAS
PubMed
Google Scholar
Timofeev, V. & Samygina, V. Protein crystallography: achievements and challenges. Crystals 13, 71 (2023).Article
CAS
Google Scholar
Platzer, G., Mayer, M., McConnell, D. B. & Konrat, R. NMR-driven structure-based drug discovery by unveiling molecular interactions. Commun. Chem. 8, 167 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Danmaliki, G. I. & Hwang, P. M. Solution NMR spectroscopy of membrane proteins. Biochim. Biophys. Acta Biomembr. 1862, 183356 (2020).Article
CAS
PubMed
Google Scholar
Hiller, S. et al. Solution structure of the integral human membrane protein VDAC-1 in detergent micelles. Science 321, 1206–1210 (2008).Article
CAS
PubMed
PubMed Central
Google Scholar
Berardi, M. J., Shih, W. M., Harrison, S. C. & Chou, J. J. Mitochondrial uncoupling protein 2 structure determined by NMR molecular fragment searching. Nature 476, 109–113 (2011).Article
CAS
PubMed
PubMed Central
Google Scholar
Mao, S. Emerging role and the signaling pathways of uncoupling protein 2 in kidney diseases. Ren. Fail. 46, 2381604 (2024).Article
PubMed
PubMed Central
Google Scholar
OuYang, B. et al. Unusual architecture of the p7 channel from hepatitis C virus. Nature 498, 521–525 (2013).Article
CAS
PubMed
PubMed Central
Google Scholar
Comarmond, C., Cacoub, P. & Saadoun, D. Treatment of chronic hepatitis C-associated cryoglobulinemia vasculitis at the era of direct-acting antivirals. Therap. Adv. Gastroenterol. 13, 1756284820942617 (2020).Article
CAS
PubMed
PubMed Central
Google Scholar
Henson, J. B. & Sise, M. E. The association of hepatitis C infection with the onset of CKD and progression into ESRD. Semin. Dial. 32, 108–118 (2019).Article
PubMed
Google Scholar
Leman, J. K. & Künze, G. Recent advances in NMR protein structure prediction with ROSETTA. Int. J. Mol. Sci. 24, 7835 (2023).Article
Google Scholar
Azinas, S. & Carroni, M. Cryo-EM uniqueness in structure determination of macromolecular complexes: a selected structural anthology. Curr. Opin. Struct. Biol. 81, 102621 (2023).Article
CAS
PubMed
Google Scholar
Piper, S. J., Johnson, R. M., Wootten, D. & Sexton, P. M. Membranes under the magnetic lens: a dive into the diverse world of membrane protein structures using cryo-EM. Chem. Rev. 122, 13989–14017 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
DeVore, K. & Chiu, P. L. Probing structural perturbation of biomolecules by extracting cryo-EM data heterogeneity. Biomolecules 12, 628 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Shen, P. S. et al. The structure of the polycystic kidney disease channel PKD2 in lipid nanodiscs. Cell 167, 763–773.e11 (2016).Article
CAS
PubMed
PubMed Central
Google Scholar
Dong, Y., Cao, L. X., Tang, H., Shi, X. Y. & He, Y. N. Structure of human M-type phospholipase A2 receptor revealed by cryo-electron microscopy. J. Mol. Biol. 429, 3825–3835 (2017).Article
CAS
PubMed
Google Scholar
Huynh, K. W. et al. Cryo-EM structure of the human SLC4A4 sodium-coupled acid-base transporter NBCe1. Nat. Commun. 9, 900 (2018).Article
PubMed
PubMed Central
Google Scholar
Zhekova, H. R. et al. Cryo-EM structures of anion exchanger 1 capture multiple states of inward- and outward-facing conformations. Commun. Biol. 5, 1372 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Stsiapanava, A. et al. Cryo-EM structure of native human uromodulin, a zona pellucida module polymer. EMBO J. 39, e106807 (2020).Article
CAS
PubMed
PubMed Central
Google Scholar
Khandelwal, N. K. et al. Structural basis of disease mutation and substrate recognition by the human SLC2A9 transporter. Proc. Natl Acad. Sci. USA 122, e2418282122 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Puri, S. et al. The Cryo-EM structure of renal amyloid fibril suggests structurally homogeneous multiorgan aggregation in AL amyloidosis. J. Mol. Biol. 435, 168215 (2023).Article
CAS
PubMed
Google Scholar
Han, B. G., Avila-Sakar, A., Remis, J. & Glaeser, R. M. Challenges in making ideal cryo-EM samples. Curr. Opin. Struct. Biol. 81, 102646 (2023).Article
CAS
PubMed
Google Scholar
Patwardhan, A., Henderson, R. & Russo, C. J. Extending the reach of single-particle cryo-EM. Curr. Opin. Struct. Biol. 92, 103005 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Liu, Y. T., Fan, H., Hu, J. J. & Zhou, Z. H. Overcoming the preferred-orientation problem in cryo-EM with self-supervised deep learning. Nat. Methods 22, 113–123 (2024).Article
PubMed
PubMed Central
Google Scholar
Wang, H., Liao, S., Yu, X., Zhang, J. & Zhou, Z. H. TomoNet: a streamlined cryogenic electron tomography software pipeline with automatic particle picking on flexible lattices. Biol. Imaging 4, e7 (2024).Article
PubMed
PubMed Central
Google Scholar
Weiss, G. L. et al. Architecture and function of human uromodulin filaments in urinary tract infections. Science 369, 1005–1010 (2020).Article
CAS
PubMed
Google Scholar
Leitner, A., Walzthoeni, T. & Aebersold, R. Lysine-specific chemical cross-linking of protein complexes and identification of cross-linking sites using LC-MS/MS and the xQuest/xProphet software pipeline. Nat. Protoc. 9, 120–137 (2014).Article
CAS
PubMed
Google Scholar
Stahl, K., Graziadei, A., Dau, T., Brock, O. & Rappsilber, J. Protein structure prediction with in-cell photo-crosslinking mass spectrometry and deep learning. Nat. Biotechnol. 41, 1810–1819 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
McCafferty, C. L., Pennington, E. L., Papoulas, O., Taylor, D. W. & Marcotte, E. M. Does AlphaFold2 model proteins’ intracellular conformations? An experimental test using cross-linking mass spectrometry of endogenous ciliary proteins. Commun. Biol. 6, 421 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Meng, Y. J. et al. Protein structure prediction via deep learning: an in-depth review. Front. Pharmacol. 16, 1498662 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Krokidis, M. G. et al. AlphaFold3: an overview of applications and performance insights. Int. J. Mol. Sci. 26, 3671 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Senior, A. W. et al. Improved protein structure prediction using potentials from deep learning. Nature 577, 706–710 (2020).Article
CAS
PubMed
Google Scholar
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Varadi, M. et al. AlphaFold protein structure database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 50, D439–D444 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Rohl, C. A., Strauss, C. E. M., Misura, K. M. S. & Baker, D. Protein structure prediction using Rosetta. Method. Enzymol. 383, 66–93 (2004).Article
CAS
Google Scholar
Leaver-Fay, A. et al. Rosetta3: an object-oriented software suite for the simulation and design of macromolecules. Meth Enzymol. 487, 545–574 (2011).Article
CAS
Google Scholar
Schmitz, C., Vernon, R., Otting, G., Baker, D. & Huber, T. Protein structure determination from pseudocontact shifts using ROSETTA. J. Mol. Biol. 416, 668–677 (2012).Article
CAS
PubMed
PubMed Central
Google Scholar
Kuenze, G., Bonneau, R., Leman, J. K. & Meiler, J. Integrative protein modelling in RosettaNMR from sparse paramagnetic restraints. Structure 27, 1721–1734.e5 (2019).Article
CAS
PubMed
PubMed Central
Google Scholar
Wang, R. Y. R. et al. De novo protein structure determination from near-atomic-resolution cryo-EM maps. Nat. Methods 12, 335–338 (2015).Article
CAS
PubMed
PubMed Central
Google Scholar
Baek, M. et al. Accurate prediction of protein structures and interactions using a three-track neural network. Science 373, 871–876 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Krishna, R. et al. Generalized biomolecular modelling and design with RoseTTAFold all-atom. Science 384, eadl2528 (2024).Article
CAS
PubMed
Google Scholar
Harmalkar, A., Lyskov, S. & Gray, J. J. Reliable protein-protein docking with AlphaFold, Rosetta, and replica exchange. Elife 13, RP94029 (2025).Article
PubMed
PubMed Central
Google Scholar
Silva, D. A. et al. De novo design of potent and selective mimics of IL-2 and IL-15. Nature 565, 186–191 (2019).Article
CAS
PubMed
PubMed Central
Google Scholar
Cao, L. X. et al. Design of protein-binding proteins from the target structure alone. Nature 605, 551–560 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Watson, J. L. et al. De novo design of protein structure and function with RFdiffusion. Nature 620, 1089–1100 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Drake, Z. C., Fowler, A. G., Blum, A. A. & Lindert, S. Enhanced protein complex prediction via Rosetta, AlphaFold, and nondifferential covalent labeling mass spectrometry. J. Phys. Chem. B 129, 6489–6497 (2025).Article
CAS
PubMed
Google Scholar
Stahl, K. et al. Modelling protein complexes with crosslinking mass spectrometry and deep learning. Nat. Commun. 15, 7866 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Manalastas-Cantos, K. et al. Modelling flexible protein structure with AlphaFold2 and crosslinking mass spectrometry. Mol. Cell Proteom. 23, 100724 (2024).Article
CAS
Google Scholar
Terwilliger, T. C. et al. Accelerating crystal structure determination with iterative prediction. Acta Crystallogr. D Struct. Biol. 79, 234–244 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Barbarin-Bocahu, I. & Graille, M. The X-ray crystallography phase problem solved thanks to AlphaFold and RoseTTAFold models: a case-study report. Acta Crystallogr. D Struct. Biol. 78, 517–531 (2022).Article
CAS
PubMed
Google Scholar
Laurents, D. V. AlphaFold 2 and NMR spectroscopy: partners to understand protein structure, dynamics and function. Front. Mol. Biosci. 9, 906437 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Tegunov, D., Xue, L., Dienemann, C., Cramer, P. & Mahamid, J. Multi-particle cryo-EM refinement with M visualizes ribosome-antibiotic complex at 3.5 Å in cells. Nat. Methods 18, 186–193 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Pettersen, E. F. et al. UCSF Chimera-a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).Article
CAS
PubMed
Google Scholar
Sanchez-Garcia, R. et al. DeepEMhancer: a deep learning solution for cryo-EM volume post-processing. Commun. Biol. 4, 874 (2021).Article
PubMed
PubMed Central
Google Scholar
Ma, X. & Si, D. Beyond current boundaries: integrating deep learning and AlphaFold for enhanced protein structure prediction from low-resolution cryo-EM maps. Comput. Biol. Chem. 119, 108494 (2025).Article
CAS
PubMed
Google Scholar
Dai, X., Wu, L., Yoo, S. & Liu, Q. Integrating AlphaFold and deep learning for atomistic interpretation of cryo-EM maps. Brief. Bioinform 24, bbad405 (2023).Article
PubMed
Google Scholar
Jamali, K. et al. Automated model building and protein identification in cryo-EM maps. Nature 628, 450–457 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Wang, J. et al. End-to-end cryo-EM complex structure determination with high accuracy and ultra-fast speed. Nat. Mach. Intell. 7, 1091–1103 (2025).Article
Google Scholar
Wohlwend, J. et al. Boltz-1 democratizing biomolecular interaction modelling. Preprint at bioRxiv https://doi.org/10.1101/2024.11.19.624167 (2024).Nussinov, R., Zhang, M. Z., Liu, Y. L. & Jang, H. AlphaFold, artificial intelligence (AI), and allostery. J. Phys. Chem. B 126, 6372–6383 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Chakravarty, D. & Porter, L. L. AlphaFold2 fails to predict protein fold switching. Protein Sci. 31, e4353 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Gu, X. Y., Aranganathan, A. & Tiwary, P. Empowering AlphaFold2 for protein conformation selective drug discovery with AlphaFold2-RAVE. Elife 13, e99702 (2024).Article
Google Scholar
Pratt, O. S. et al. AlphaFold 2, but not AlphaFold 3, predicts confident but unrealistic R-solenoid structures for repeat proteins. Comput. Struct. Biotec 27, 467–477 (2025).Article
CAS
Google Scholar
Terwilliger, T. C. et al. AlphaFold predictions are valuable hypotheses and accelerate but do not replace experimental structure determination. Nat. Methods 21, 110–116 (2024).Article
CAS
PubMed
Google Scholar
Krokidis, M. G., Dimitrakopoulos, G. N., Vrahatis, A. G., Exarchos, T. P. & Vlamos, P. Challenges and limitations in computational prediction of protein misfolding in neurodegenerative diseases. Front. Comput. Neurosc 17, 1323182 (2024).Article
Google Scholar
Evans, R. et al. Protein complex prediction with AlphaFold-Multimer. Preprint at bioRxiv https://doi.org/10.1101/2021.10.04.463034 (2021).Akdel, M. et al. A structural biology community assessment of AlphaFold2 applications. Nat. Struct. Mol. Biol. 29, 1056–1067 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Buel, G. R. & Walters, K. J. Can AlphaFold2 predict the impact of missense mutations on structure? Nat. Struct. Mol. Biol. 29, 1–2 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Pak, M. A. et al. Using AlphaFold to predict the impact of single mutations on protein stability and function. PLoS One 18, e0282689 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Ruff, K. M. & Pappu, R. V. AlphaFold and implications for intrinsically disordered proteins. J. Mol. Biol. 433, 167208 (2021).Article
CAS
PubMed
Google Scholar
Lin, Z. M. et al. Evolutionary-scale prediction of atomic-level protein structure with a language model. Science 379, 1123–1130 (2023).Article
CAS
PubMed
Google Scholar
Wu, R. et al. High-resolution de novo structure prediction from primary sequence. Preprint at bioRxiv https://doi.org/10.1101/2022.07.21.500999 (2022).Cheng, J. et al. Accurate proteome-wide missense variant effect prediction with AlphaMissense. Science 381, 1303 (2023).Article
Google Scholar
van Kempen, M. et al. Fast and accurate protein structure search with Foldseek. Nat. Biotechnol. 42, 184–193 (2024).
Google Scholar
Passaro, S. et al. Boltz-2: towards accurate and efficient binding affinity prediction. Preprint at bioRxiv https://doi.org/10.1101/2025.06.14.659707 (2025).Chen, Y. H., Xu, Y. X., Liu, D., Xing, Y. G. & Gong, H. P. An end-to-end framework for the prediction of protein structure and fitness from single sequence. Nat. Commun. 15, 7400 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Fang, X. et al. HelixFold-Multimer: elevating protein complex structure prediction to new heights. Preprint at arXiv https://arxiv.org/abs/2404.10260 (2024).Jing, B. et al. EigenFold: generative protein structure prediction with diffusion models. In ICLR 2023 Machine Learning for Drug Discovery Workshop (ICLR, 2023).Ruffolo, J. A., Chu, L. S., Mahajan, S. P. & Gray, J. J. Fast, accurate antibody structure prediction from deep learning on massive set of natural antibodies. Nat. Commun. 14, 38063 (2023).Article
Google Scholar
Wang, Y. et al. xTrimoABFold: improving antibody structure prediction without multiple sequence alignments. Preprint at arXiv https://arxiv.org/abs/2212.00735 (2022).Qiao, Z. et al. NeuralPLexer3: accurate biomolecular complex structure prediction with flow models. In Thirty-ninth Annual Conference on Neural Information Processing Systems (NeurIPS, 2025).Ng, P. C. & Henikoff, S. SIFT: predicting amino acid changes that affect protein function. Nucleic Acids Res. 31, 3812–3814 (2003).Article
CAS
PubMed
PubMed Central
Google Scholar
Adzhubei, I. A. et al. A method and server for predicting damaging missense mutations. Nat. Methods 7, 248–249 (2010).Article
CAS
PubMed
PubMed Central
Google Scholar
Medaer, L. et al. Residual cystine transport activity for specific infantile and juvenile CTNS mutations in a PTEC-based addback model. Cells 13, 646 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Gao, H. et al. The landscape of tolerated genetic variation in humans and primates. Science 380, eabn8197 (2023).Article
Google Scholar
Rives, A. et al. Biological structure and function emerge from scaling unsupervised learning to 250 million protein sequences. Proc. Natl Acad. Sci. USA 118, e2016239118 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Livesey, B. J. & Marsh, J. A. Interpreting protein variant effects with computational predictors and deep mutational scanning. Dis. Model. Mech. 15, dmm049510 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Stein, D. et al. Genome-wide prediction of pathogenic gain- and loss-of-function variants from ensemble learning of a diverse feature set. Genome Med. 15, 103 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Tunyasuvunakool, K. et al. Highly accurate protein structure prediction for the human proteome. Nature 596, 590–596 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Khan, A. et al. Metabolic gene function discovery platform GeneMAP identifies SLC25A48 as necessary for mitochondrial choline import. Nat. Genet. 56, 1614–1623 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Maity, S. & Sharma, K. The mitochondrial choline transporter, SLC25A48, regulates urine and blood choline levels in humans. Kidney Int. 107, 225–227 (2025).Article
CAS
PubMed
Google Scholar
Patil, S. et al. The membrane transporter SLC25A48 enables transport of choline into human mitochondria. Kidney Int. 107, 296–301 (2025).Article
CAS
PubMed
Google Scholar
Reithmeier, R. A. et al. Band 3, the human red cell chloride/bicarbonate anion exchanger (AE1, SLC4A1), in a structural context. Biochim. Biophys. Acta 1858, 1507–1532 (2016).Article
CAS
PubMed
Google Scholar
Essuman, G. et al. SLC4A1 mutations that cause distal renal tubular acidosis alter cytoplasmic pH and cellular autophagy. Elife 13, RP108253 (2024).
Google Scholar
Peng, S. Q. et al. A gain-of-function mutation in ATP6V0A4 drives primary distal renal tubular alkalosis with enhanced V-ATPase activity. J. Clin. Invest. 135, e188807 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Subramanya, A. R. & Ellison, D. H. Distal convoluted tubule. Clin. J. Am. Soc. Nephrol. 9, 2147–2163 (2014).Article
CAS
PubMed
PubMed Central
Google Scholar
Blanchard, A. et al. Gitelman syndrome: consensus and guidance from a kidney disease: improving global outcomes (KDIGO) controversies conference. Kidney Int. 91, 24–33 (2017).Article
PubMed
Google Scholar
Nan, J. et al. Cryo-EM structure of the human sodium-chloride cotransporter NCC. Sci. Adv. 8, eadd7176 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Fan, M. R., Zhang, J. X., Lee, C. L., Zhang, J. R. & Feng, L. Structure and thiazide inhibition mechanism of the human Na-Cl cotransporter. Nature 614, 788–793 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Li, N. & Gu, H. F. Genetic and biological effects of SLC12A3, a sodium-chloride cotransporter, in Gitelman syndrome and diabetic kidney disease. Front. Genet. 13, 799224 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Ji, X. C., Zhao, N., Liu, H. X., Wu, Y. T. & Liu, L. C. Case report: two novel compound heterozygous variant of SLC12A3 gene in a Gitelman syndrome family and literature review. Front. Genet. 15, 1391015 (2024).Article
PubMed
PubMed Central
Google Scholar
Wang, N. et al. Functional evaluation of novel compound heterozygous variants in SLC12A3 of Gitelman syndrome. Orphanet J. Rare Dis. 20, 66 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Welling, P. A. & Ho, K. A comprehensive guide to the ROMK potassium channel: form and function in health and disease. Am. J. Physiol. Renal Physiol. 297, F849–F863 (2009).Article
CAS
PubMed
PubMed Central
Google Scholar
Nguyen, N. H. et al. Genome mining yields putative disease-associated ROMK variants with distinct defects. PLoS Genet. 19, e1011051 (2023).Article
PubMed
PubMed Central
Google Scholar
Nguyen, N. H. et al. Characterization of hyperactive mutations in the renal potassium channel ROMK uncovers unique effects on channel biogenesis and ion conductance. Mol. Biol. Cell 35, ar119 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Hosoyamada, M., Sekine, T., Kanai, Y. & Endou, H. Molecular cloning and functional expression of a multispecific organic anion transporter from human kidney. Am. J. Physiol. Renal Physiol. 276, F122–F128 (1999).Article
CAS
Google Scholar
Morrissey, K. M., Stocker, S. L., Wittwer, M. B., Xu, L. & Giacomini, K. M. Renal transporters in drug development. Annu. Rev. Pharmacol. Toxicol. 53, 503–529 (2013).Article
CAS
PubMed
Google Scholar
Riedmaier, A. E., Nies, A. T., Schaeffeler, E. & Schwab, M. Organic anion transporters and their implications in pharmacotherapy. Pharmacol. Rev. 64, 421–449 (2012).Article
CAS
Google Scholar
Parker, J. L., Kato, T., Kuteyi, G., Sitsel, O. & Newstead, S. Molecular basis for selective uptake and elimination of organic anions in the kidney by OAT1. Nat. Struct. Mol. Biol. 30, 1786–1793 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Akunjee, M. M., Khosla, S. G., Nylen, E. S. & Sen, S. SGLT2 inhibitors use in kidney disease: what did we learn? Am. J. Physiol. Endocrinol. Metab. 328, E856–E868 (2025).Article
CAS
PubMed
Google Scholar
Hiraizumi, M. et al. Transport and inhibition mechanism of the human SGLT2-MAP17 glucose transporter. Nat. Struct. Mol. Biol. 31, 159–169 (2024).Article
CAS
PubMed
Google Scholar
Jamalpoor, A., Othman, A., Levtchenko, E. N., Masereeuw, R. & Janssen, M. J. Molecular mechanisms and treatment options of nephropathic cystinosis. Trends Mol. Med. 27, 673–686 (2021).Article
CAS
PubMed
Google Scholar
Guo, X. et al. Structure and mechanism of human cystine exporter cystinosin. Cell 185, 3739–3752.e18 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Lobel, M. et al. Structural basis for proton coupled cystine transport by cystinosin. Nat. Commun. 13, 4845 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Schaub, C. et al. Cation channel conductance and pH gating of the innate immunity factor APOL1 are governed by pore-lining residues within the C-terminal domain. J. Biol. Chem. 295, 13138–13149 (2020).Article
CAS
PubMed
PubMed Central
Google Scholar
Tabachnikov, O., Skorecki, K. & Kruzel-Davila, E. APOL1 nephropathy — a population genetics success story. Curr. Opin. Nephrol. Hypertens. 33, 447–455 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Madhavan, S. M., Hansen, A. L., Cao, S., Sedor, J. R. & Buck, M. Towards the NMR solution structure and the dynamics of the C-terminal region of APOL1 and its G1, G2 variants with a membrane mimetic. Preprint at bioRxiv https://doi.org/10.1101/2021.03.16.435683 (2021).Kulkarni, K. & Hussain, T. Megalin: a sidekick or nemesis of the kidney? J. Am. Soc. Nephrol. 36, 293–300 (2025).Article
PubMed
Google Scholar
Charlton, J. R. et al. Beyond the tubule: pathological variants of LRP2, encoding the megalin receptor, result in glomerular loss and early progressive chronic kidney disease. Am. J. Physiol. Renal Physiol. 319, F988–F999 (2020).Article
CAS
PubMed
PubMed Central
Google Scholar
Beenken, A. et al. Structures of LRP2 reveal a molecular machine for endocytosis. Cell 186, 821–836.e13 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Chebib, F. T., Hanna, C., Harris, P. C., Torres, V. E. & Dahl, N. K. Autosomal dominant polycystic kidney disease: a review. JAMA 333, 1708–1719 (2025).Article
CAS
PubMed
Google Scholar
Palomero, E. O., Larmore, M. & DeCaen, P. G. Polycystin channel complexes. Annu. Rev. Physiol. 85, 425–448 (2023).Article
Google Scholar
Palomero, O. E., Guadarrama, E. & DeCaen, P. G. Pathogenic variants in the polycystin pore helix cause distinct forms of channel dysfunction. Proc. Natl Acad. Sci. USA 122, e2421362122 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Ng, L. C. T., Vien, T. N., Yarov-Yarovoy, V. & DeCaen, P. G. Opening TRPP2 (PKD2L1) requires the transfer of gating charges. Proc. Natl Acad. Sci. USA 116, 15540–15549 (2019).Article
CAS
PubMed
PubMed Central
Google Scholar
Su, Q. et al. Structure of the human PKD1-PKD2 complex. Science 361, eaat9819 (2018).Article
PubMed
Google Scholar
Lemoine, H. et al. Monoallelic pathogenic ALG5 variants cause atypical polycystic kidney disease and interstitial fibrosis. Am. J. Hum. Genet. 109, 1484–1499 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Elhassan, E. A. E. et al. A novel monoallelic ALG5 variant causing late-onset ADPKD and tubulointerstitial fibrosis. Kidney Int. Rep. 9, 2209–2226 (2024).Article
PubMed
PubMed Central
Google Scholar
von Schnakenburg, C., Fliegauf, M. & Omran, H. Nephrocystin and ciliary defects not only in the kidney? Pediatr. Nephrol. 22, 765–769 (2007).Article
Google Scholar
Saunier, S. et al. Characterization of the NPHP1 locus: mutational mechanism involved in deletions in familial juvenile nephronophthisis. Am. J. Hum. Genet. 66, 778–789 (2000).Article
CAS
PubMed
PubMed Central
Google Scholar
Caridi, G. et al. Stop codon at arginine 586 is the prevalent nephronopthisis type 1 mutation in Italy. Nephrol. Dial. Transpl. 21, 2301–2303 (2006).Article
Google Scholar
Leggatt, G. et al. A genotype-to-phenotype approach suggests under-reporting of single nucleotide variants in nephrocystin-1 (NPHP1) related disease (UK 100,000 Genomes Project). Sci. Rep. 13, 32169 (2023).Article
Google Scholar
Reiter, J. F. & Leroux, M. R. Genes and molecular pathways underpinning ciliopathies. Nat. Rev. Mol. Cell Biol. 18, 533–547 (2017).Article
CAS
PubMed
PubMed Central
Google Scholar
Ishikawa, H. & Marshall, W. F. Ciliogenesis: building the cell’s antenna. Nat. Rev. Mol. Cell Biol. 12, 222–234 (2011).Article
CAS
PubMed
Google Scholar
Jordan, M. A., Diener, D. R., Stepanek, L. & Pigino, G. The cryo-EM structure of intraflagellar transport trains reveals how dynein is inactivated to ensure unidirectional anterograde movement in cilia. Nat. Cell Biol. 20, 1250–1255 (2018).Article
CAS
PubMed
Google Scholar
McCafferty, C. L. et al. Integrative modelling reveals the molecular architecture of the intraflagellar transport A (IFT-A) complex. Elife 11, e81977 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Meleppattu, S., Zhou, H. X., Dai, J., Gui, M. & Brown, A. Mechanism of IFT-A polymerization into trains for ciliary transport. Cell 185, 4986–4998.e12 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Hesketh, S. J., Mukhopadhyay, A. G., Nakamura, D., Toropova, K. & Roberts, A. J. IFT-A structure reveals carriages for membrane protein transport into cilia. Cell 185, 4971–4985.e16 (2022).Article
CAS
PubMed
Google Scholar
Lacey, S. E., Foster, H. E. & Pigino, G. The molecular structure of IFT-A and IFT-B in anterograde intraflagellar transport trains. Nat. Struct. Mol. Biol. 30, 584–593 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Ma, Y. Y. et al. Structural insight into the intraflagellar transport complex IFT-A and its assembly in the anterograde IFT train. Nat. Commun. 14, 1506 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Senum, S. R. et al. Monoallelic IFT140 pathogenic variants are an important cause of the autosomal dominant polycystic kidney-spectrum phenotype. Am. J. Hum. Genet. 109, 136–156 (2022).Article
CAS
PubMed
Google Scholar
Jiang, M. et al. Human IFT-A complex structures provide molecular insights into ciliary transport. Cell Res. 33, 288–298 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Kume, T., Deng, K. & Hogan, B. L. Minimal phenotype of mice homozygous for a null mutation in the forkhead/winged helix gene, Mf2. Mol. Cell Biol. 20, 1419–1425 (2000).Article
CAS
PubMed
PubMed Central
Google Scholar
Riedhammer, K. M. et al. Implication of transcription factor FOXD2 dysfunction in syndromic congenital anomalies of the kidney and urinary tract (CAKUT). Kidney Int. 105, 844–864 (2024).Article
CAS
PubMed
Google Scholar
Kakun, R. R., Melamed, Z. & Perets, R. PAX8 in the junction between development and tumorigenesis. Int. J. Mol. Sci. 23, 7410 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Li, L., Hossain, S. M. & Eccles, M. R. The role of the PAX genes in renal cell carcinoma. Int. J. Mol. Sci. 25, 6730 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Caliskan, A., Gulfidan, G., Sinha, R. & Arga, K. Y. Differential interactome proposes subtype-specific biomarkers and potential therapeutics in renal cell carcinomas. J. Pers. Med. 11, 158 (2021).Article
PubMed
PubMed Central
Google Scholar
Pei, J., Zhang, J. & Cong, Q. Computational analysis of protein-protein interactions of cancer drivers in renal cell carcinoma. FEBS Open. Bio 14, 112–126 (2024).Article
CAS
PubMed
Google Scholar
Goldsmith, E. J. & Rodan, A. R. Intracellular ion control of WNK signaling. Annu. Rev. Physiol. 85, 383–406 (2023).Article
CAS
PubMed
Google Scholar
Cornelius, R. J., Maeoka, Y., Shinde, U. & McCormick, J. A. Familial hyperkalemic hypertension. Compr. Physiol. 14, 5839–5874 (2024).Article
PubMed
PubMed Central
Google Scholar
Faezov, B. & Dunbrack, R. L. AlphaFold2 models of the active form of all 437 catalytically competent human protein kinase domains. Preprint at bioRxiv https://doi.org/10.1101/2023.07.21.550125 (2023).Pei, J. & Cong, Q. Computational analysis of regulatory regions in human protein kinases. Protein Sci. 32, e4764 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Amnekar, R. V. et al. NRBP1 pseudokinase binds to and activates the WNK pathway in response to osmotic stress. Sci. Adv. 11, eadv4636 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Puapatanakul, P. & Miner, J. H. Alport syndrome and Alport kidney diseases — elucidating the disease spectrum. Curr. Opin. Nephrol. Hypertens. 33, 283–290 (2024).Article
PubMed
PubMed Central
Google Scholar
Al-Shaer, A. et al. Sequence-dependent mechanics of collagen reflect its structural and functional organization. Biophys. J. 120, 4013–4028 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Chen, D. et al. Effects of a novel COL4A3 homozygous/heterozygous splicing mutation on the mild phenotype in a family with autosomal recessive Alport syndrome and a literature review. Mol. Genet. Genomic Med. 13, e70053 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Chen, S., Zhang, Y., He, J. & Yang, D. Case report: a novel compound heterozygous variant in the COL4A3 gene was identified in a patient with autosomal recessive Alport syndrome. Front. Genet. 15, 1426806 (2024).Article
PubMed
PubMed Central
Google Scholar
Kocylowski, M. K. et al. A slit-diaphragm-associated protein network for dynamic control of renal filtration. Nat. Commun. 13, 6446 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Yu, S. M., Nissaisorakarn, P., Husain, I. & Jim, B. Proteinuric Kidney diseases: a podocyte’s slit diaphragm and cytoskeleton approach. Front. Med. 5, 221 (2018).Article
Google Scholar
Padmanaban, H. K., Bheemireddy, S., Mulukala, S. K., Dunna, N. R. & Pasupulati, A. K. Structural conformations of intrinsically disordered proteins of podocyte slit-diaphragm. Comput. Struct. Biotechnol. Rep. 2, 100060 (2025).
Google Scholar
Birtasu, A. N. et al. The kidney slit diaphragm resembles a fishnet. Kidney Int. 108, 1045–1056 (2025).Article
PubMed
Google Scholar
Singh, N., Nainani, N., Arora, P. & Venuto, R. C. CKD in MYH9-related disorders. Am. J. Kidney Dis. 54, 732–740 (2009).Article
CAS
PubMed
Google Scholar
Tabibzadeh, N. et al. MYH9-related disorders display heterogeneous kidney involvement and outcome. Clin. Kidney J. 12, 494–502 (2019).Article
CAS
PubMed
Google Scholar
Freeman, N. S. et al. Familial idiopathic glomerular disease due to a unique renal-predominant phenotype of MYH9-related disease: a case report. Glomerular Dis. 5, 243–249 (2025).Article
PubMed
PubMed Central
Google Scholar
Thielemans, R. et al. Unveiling the hidden power of uromodulin: a promising potential biomarker for kidney diseases. Diagnostics 13, 3077 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Olinger, E. et al. Clinical and genetic spectra of autosomal dominant tubulointerstitial kidney disease due to mutations in UMOD and MUC1. Kidney Int. 98, 717–731 (2020).Article
CAS
PubMed
Google Scholar
Stsiapanava, A. et al. Structure of the decoy module of human glycoprotein 2 and uromodulin and its interaction with bacterial adhesin FimH. Nat. Struct. Mol. Biol. 29, 190–193 (2022).Article
CAS
PubMed
PubMed Central
Google Scholar
Bogdan, R. G. et al. Atypical hemolytic uremic syndrome: a review of complement dysregulation, genetic susceptibility and multiorgan involvement. J. Clin. Med. 14, 2527 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Cao, W., Liu, Y., Zhang, X. F. & Zheng, X. L. A mutant complement factor H (W1183R) enhances proteolytic cleavage of von Willebrand factor by ADAMTS-13 under shear. J. Thromb. Haemost. 23, 1229–1240 (2025).Article
CAS
PubMed
Google Scholar
Xi, K. et al. Unveiling the mechanisms of nephrotoxicity caused by nephrotoxic compounds using toxicological network analysis. Mol. Ther. Nucleic Acids 34, 102075 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Shimokawa, M. et al. Acute tubular injury and Fanconi Syndrome associated with red yeast rice supplement. Kidney Int. Rep. 10, 956–959 (2025).Article
PubMed
Google Scholar
Hayama, T., Sugawara, R., Kamata, R., Sekijima, M. & Takeda, K. Comprehensive molecular docking on the AlphaFold-predicted protein structure proteome: identifying target protein candidates for puberulic acid. J. Toxicol. Sci. 50, 309–324, https://doi.org/10.2131/jts.50.309 (2025).Article
CAS
PubMed
Google Scholar
Lely, A. T., Hamming, I., van Goor, H. & Navis, G. J. Renal ACE2 expression in human kidney disease. J. Pathol. 204, 587–593 (2004).Article
CAS
PubMed
Google Scholar
Bell, S., Perkins, G. B., Anandh, U. & Coates, P. T. COVID and the kidney: an update. Semin. Nephrol. 43, 151471 (2023).Article
CAS
PubMed
Google Scholar
Kilim, O., Mentes, A., Pal, B., Csabai, I. & Gellert, A. SARS-CoV-2 receptor-binding domain deep mutational AlphaFold2 structures. Sci. Data 10, 134 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Raisinghani, N., Alshahrani, M., Gupta, G. & Verkhivker, G. AlphaFold2 modelling and molecular dynamics simulations of the conformational ensembles for the SARS-CoV-2 spike Omicron JN.1, KP.2 and KP.3 variants: mutational profiling of binding energetics reveals epistatic drivers of the ACE2 affinity and escape hotspots of antibody resistance. Viruses 16, 1458 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar
Flower, T. G. & Hurley, J. H. Crystallographic molecular replacement using an in silico-generated search model of SARS-CoV-2 ORF8. Protein Sci. 30, 728–734 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Robertson, A. J., Courtney, J. M., Shen, Y., Ying, J. & Bax, A. Concordance of X-ray and AlphaFold2 models of SARS-CoV-2 main protease with residual dipolar couplings measured in solution. J. Am. Chem. Soc. 143, 19306–19310 (2021).Article
CAS
PubMed
PubMed Central
Google Scholar
Trevino, M. A., Pantoja-Uceda, D., Laurents, D. V. & Mompean, M. SARS-CoV-2 Nsp8 N-terminal domain folds autonomously and binds dsRNA. Nucleic Acids Res. 51, 10041–10048 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Thakkar, R. et al. De novo design of a stapled peptide targeting SARS-CoV-2 spike protein receptor-binding domain. RSC Med. Chem. 14, 1722–1733 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Hoste, E. A. J. et al. Global epidemiology and outcomes of acute kidney injury. Nat. Rev. Nephrol. 14, 607–625 (2018).Article
CAS
PubMed
Google Scholar
Yang, L. et al. KIM-1-mediated phagocytosis reduces acute injury to the kidney. J. Clin. Invest. 125, 1620–1636 (2015).Article
PubMed
PubMed Central
Google Scholar
Yang, C. et al. Kidney injury molecule-1 is a potential receptor for SARS-CoV-2. J. Mol. Cell Biol. 13, 185–196 (2021).Article
PubMed
PubMed Central
Google Scholar
Xiao, Y. et al. A rationally designed injury kidney targeting peptide library and its application in rescuing acute kidney injury. Sci. Adv. 11, eadt3943 (2025).Article
CAS
PubMed
PubMed Central
Google Scholar
Wagner, F. R. et al. Preparing samples from whole cells using focused-ion-beam milling for cryo-electron tomography. Nat. Protoc. 15, 2041–2070 (2020).Article
CAS
PubMed
PubMed Central
Google Scholar
Chen, Z. et al. De novo protein identification in mammalian sperm using cryoelectron tomography and AlphaFold2 docking. Cell 186, 5041–5053 (2023).Article
CAS
PubMed
PubMed Central
Google Scholar
Pierson, J. A., Yang, J. E. & Wright, E. R. Recent advances in correlative cryo-light and electron microscopy. Curr. Opin. Struct. Biol. 89, 102934 (2024).Article
CAS
PubMed
PubMed Central
Google Scholar