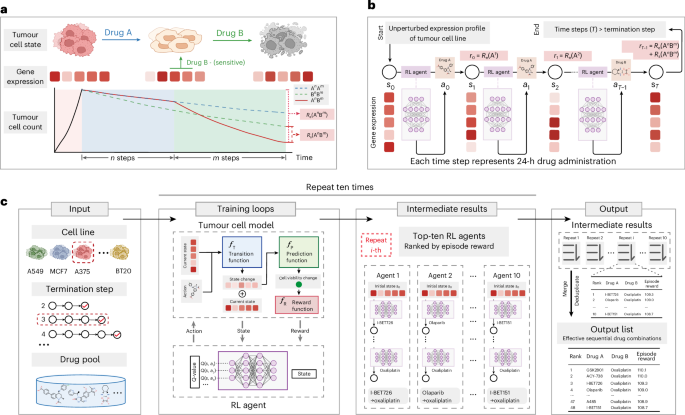
Summarize this content to 100 words: Parreno, V. et al. Transient loss of polycomb components induces an epigenetic cancer fate. Nature 629, 688–696 (2024).Article
Google Scholar
Brown, R., Curry, E., Magnani, L., Wilhelm-Benartzi, C. S. & Borley, J. Poised epigenetic states and acquired drug resistance in cancer. Nat. Rev. Cancer 14, 747–753 (2014).Article
Google Scholar
Adduri, A. K. et al. Predicting cellular responses to perturbation across diverse contexts with State. Preprint at bioRxiv https://doi.org/10.1101/2025.06.26.661135 (2025).Zhao, S. et al. SToFM: a multi-scale foundation model for spatial transcriptomics. Preprint at http://arxiv.org/abs/2507.11588 (2025).Bunne, C. et al. How to build the virtual cell with artificial intelligence: priorities and opportunities. Cell 187, 7045–7063 (2024).Article
Google Scholar
Salvador-Barbero, B. et al. CDK4/6 inhibitors impair recovery from cytotoxic chemotherapy in pancreatic adenocarcinoma. Cancer Cell 37, 340–353.e346 (2020).Article
Google Scholar
Fang, Y. et al. Sequential therapy with PARP and WEE1 inhibitors minimizes toxicity while maintaining efficacy. Cancer Cell 35, 851–867.e857 (2019).Article
Google Scholar
Lee, M. J. et al. Sequential application of anticancer drugs enhances cell death by rewiring apoptotic signaling networks. Cell 149, 780–794 (2012).Article
Google Scholar
Goldman, A. et al. Temporally sequenced anticancer drugs overcome adaptive resistance by targeting a vulnerable chemotherapy-induced phenotypic transition. Nat. Commun. 6, 6139 (2015).Article
Google Scholar
Liu, Z. et al. A proteomic and phosphoproteomic landscape of KRAS mutant cancers identifies combination therapies. Mol. Cell 81, 4076–4090.e4078 (2021).Article
Google Scholar
Wang, L., Lankhorst, L. & Bernards, R. Exploiting senescence for the treatment of cancer. Nat. Rev. Cancer 22, 340–355 (2022).Article
Google Scholar
Wang, C. et al. Inducing and exploiting vulnerabilities for the treatment of liver cancer. Nature 574, 268–272 (2019).Article
Google Scholar
Li, F. et al. Blocking methionine catabolism induces senescence and confers vulnerability to GSK3 inhibition in liver cancer. Nat. Cancer 5, 131–146 (2024).Silver, D. et al. Mastering the game of Go with deep neural networks and tree search. Nature 529, 484–489 (2016).Article
Google Scholar
Baek, M. et al. Accurate prediction of protein structures and interactions using a three-track neural network. Science 373, 871–876 (2021).Article
Google Scholar
Wang, G. et al. Optimized glycemic control of type 2 diabetes with reinforcement learning: a proof-of-concept trial. Nat. Med. 29, 2633–2642 (2023).Article
Google Scholar
Yala, A. et al. Optimizing risk-based breast cancer screening policies with reinforcement learning. Nat. Med. 28, 136–143 (2022).Article
Google Scholar
Zhang, H. et al. Algorithm for optimized mRNA design improves stability and immunogenicity. Nature 621, 396–403 (2023).Article
Google Scholar
Weaver, D. T., King, E. S., Maltas, J. & Scott, J. G. Reinforcement learning informs optimal treatment strategies to limit antibiotic resistance. Proc. Natl Acad. Sci. USA 121, e2303165121 (2024).Article
Google Scholar
Jo, K., Sung, I., Lee, D., Jang, H. & Kim, S. Inferring transcriptomic cell states and transitions only from time series transcriptome data. Sci. Rep. 11, 12566 (2021).Article
Google Scholar
Rockne, R. C. et al. State-transition analysis of time-sequential gene expression identifies critical points that predict development of acute myeloid leukemia. Cancer Res. 80, 3157–3169 (2020).Article
Google Scholar
Mnih, V. et al. Human-level control through deep reinforcement learning. Nature 518, 529–533 (2015).Article
Google Scholar
Lotfollahi, M. et al. Predicting cellular responses to complex perturbations in high-throughput screens. Mol. Syst. Biol. 19, e11517 (2023).Article
Google Scholar
Huang, W. & Liu, H. Predicting single-cell cellular responses to perturbations using cycle consistency learning. Bioinformatics 40, i462–i470 (2024).Article
Google Scholar
Piran, Z., Cohen, N., Hoshen, Y. & Nitzan, M. Disentanglement of single-cell data with biolord. Nat. Biotechnol. 42, 1678–1683 (2024).Article
Google Scholar
Qi, X. et al. Predicting transcriptional responses to novel chemical perturbations using deep generative model for drug discovery. Nat. Commun. 15, 9256 (2024).Article
Google Scholar
Lotfollahi, M., Wolf, F. A. & Theis, F. J. scGen predicts single-cell perturbation responses. Nat. Methods 16, 715–721 (2019).Article
Google Scholar
Szalai, B. et al. Signatures of cell death and proliferation in perturbation transcriptomics data—from confounding factor to effective prediction. Nucleic Acids Res. 47, 10010–10026 (2019).Article
Google Scholar
Patwardhan, G. A. et al. Treatment scheduling effects on the evolution of drug resistance in heterogeneous cancer cell populations. NPJ Breast Cancer 7, 60 (2021).Article
Google Scholar
Johnson, T. I. et al. Quantifying cell cycle-dependent drug sensitivities in cancer using a high throughput synchronisation and screening approach. EBioMedicine 68, 103396 (2021).Article
Google Scholar
Vijayaraghavalu, S., Dermawan, J. K., Cheriyath, V. & Labhasetwar, V. Highly synergistic effect of sequential treatment with epigenetic and anticancer drugs to overcome drug resistance in breast cancer cells is mediated via activation of p21 gene expression leading to G2/M cycle arrest. Mol. Pharm. 10, 337–352 (2013).Article
Google Scholar
Easwaran, H., Tsai, H.-C. & Baylin, S. B. Cancer epigenetics: tumor heterogeneity, plasticity of stem-like states, and drug resistance. Mol. Cell 54, 716–727 (2014).Article
Google Scholar
Bloom, S. I., Islam, M. T., Lesniewski, L. A. & Donato, A. J. Mechanisms and consequences of endothelial cell senescence. Nat. Rev. Cardiol. 20, 38–51 (2023).Article
Google Scholar
Sutton, R. S. & Barto, A. G. Reinforcement Learning: An Introduction (MIT Press, 2018).Subramanian, A. et al. A next generation connectivity map: L1000 platform and the first 1,000,000 profiles. Cell 171, 1437–1452. e1417 (2017).Article
Google Scholar
Barrett, T. et al. NCBI GEO: archive for functional genomics data sets—update. Nucleic Acids Res. 41, D991–D995 (2012).Article
Google Scholar
RDKit: Open-Source Cheminformatics; https://www.rdkit.orgEnache, O. M. et al. The GCTx format and cmap {Py, R, M, J} packages: resources for optimized storage and integrated traversal of annotated dense matrices. Bioinformatics 35, 1427–1429 (2019).Article
Google Scholar
Kingma, D. P. & Ba, J. Adam: a method for stochastic optimization. Preprint at http://arxiv.org/abs/1412.6980 (2014).Prechelt, L. in Neural Networks: Tricks of the Trade 55–69 (Springer, 2002).Paszke, A. et al. PyTorch: an imperative style, high-performance deep learning library. In 33rd Conference on Neural Information Processing Systems (NeurIPS 2019) https://proceedings.neurips.cc/paper_files/paper/2019/file/bdbca288fee7f92f2bfa9f7012727740-Paper.pdf (2019).Seashore-Ludlow, B. et al. Harnessing connectivity in a large-scale small-molecule sensitivity dataset. Cancer Discov. 5, 1210–1223 (2015).Article
Google Scholar
Pedregosa, F. et al. Scikit-learn: machine learning in Python. J. Mach. Learn. Res. 12, 2825–2830 (2011).MathSciNet
Google Scholar
Brownlee, J. A gentle introduction to the rectified linear unit (ReLU). Mach. Learn. Mastery 6 (2019).Yang, L., Liu, S., Tsoka, S. & Papageorgiou, L. G. Mathematical programming for piecewise linear regression analysis. Expert Syst. Appl. 44, 156–167 (2016).Article
Google Scholar
Wagenmakers, E.-J. & Farrell, S. AIC model selection using Akaike weights. Psychon. Bull. Rev. 11, 192–196 (2004).Article
Google Scholar
Wu, T. et al. clusterProfiler 4.0: a universal enrichment tool for interpreting omics data. Innovation 2, 100141 (2021).Wishart, D. S. et al. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 46, D1074–D1082 (2018).Article
Google Scholar
Virtanen, P. et al. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat. Methods 17, 261–272 (2020).Article
Google Scholar
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).Article
Google Scholar
Dobin, A. & Gingeras, T. R. Mapping RNA-seq reads with STAR. Curr. Protoc. Bioinform. 51, 11.14. 11–11.14. 19 (2015).Article
Google Scholar
Li, B. & Dewey, C. N. RSEM: accurate transcript quantification from RNA-seq data with or without a reference genome. BMC Bioinf. 12, 323 (2011).Article
Google Scholar
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).Article
Google Scholar
Brockman, G. et al. OpenAI Gym. Preprint at http://arxiv.org/abs/1606.01540 (2016).Weng, J. et al. Tianshou: a highly modularized deep reinforcement learning library. J. Mach. Learn. Res. 23, 1–6 (2022).MathSciNet
Google Scholar